
Botulinum toxin A is a neurotoxin (nerve poison), which is formed by the bacterium Clostridium botulinum. It was first identified in 1946 by Edward J. Schantz in the United States in patients with severe food poisoning. As the food poison was observed on rotten sausage, the disease was called botulism (botulus = sausage). Botulinum toxin A was first used by the ophthalmologist Alan Scott for the treatment of squinting (strabismus) in 1980. This led to the rapid expansion of the therapeutic applications of botulinum toxin A. Furthermore, botulinum toxin A was approved by the FDA (Food and Drug Administration) for 3 approved treatments:
- Neurological disorders (e.g. strabismus, eye muscle spasm, muscular torticollis)
- Aesthetic correction of facial wrinkles on the forehead.
- Treatment of overactive sweat glands in the armpits (axillary hyperhidrosis).
The use of botulinum toxin A for the treatment of facial wrinkles (forehead wrinkles, frown lines, laugh lines) was first reported in 1990. It has now become the most common cosmetic procedure in the United States.

Botox: information versus promotion
- The Swiss agency for the authorisation and supervision of therapeutic products (Swissmedic) regulates by guidelines the use of botulinum toxin in cosmetic medicine.
- The Federal Administrative Court supports a decision by Swissmedic to ban the advertisement of medical products.
- According to the legislation, the term “botox” may not be used to identify any specific preparation containing botulinum toxin A, as their naming qualifies as the unlawful advertising of prescription drugs.
- Although the term “botox” is widely used synonymous with the active substance botulinum toxin A, in order to observe Swiss regulations, we only use the neutral term “botulinum toxin A” on this website.
How does botulinum toxin A work?
Botulinum toxin A binds to receptors of nerve endings and blocks the release of the neurotransmitter acetylcholine, resulting in the temporary degradation of the effector organ. Acetylcholine is the common neurotransmitter at the nerve endings of muscles and sweat glands. Hence, botulinum toxin A can be used to weaken mimic muscles in the treatment of facial wrinkles as well as to reduce the secretion of sweat glands in the treatment of axillary hyperhidrosis.
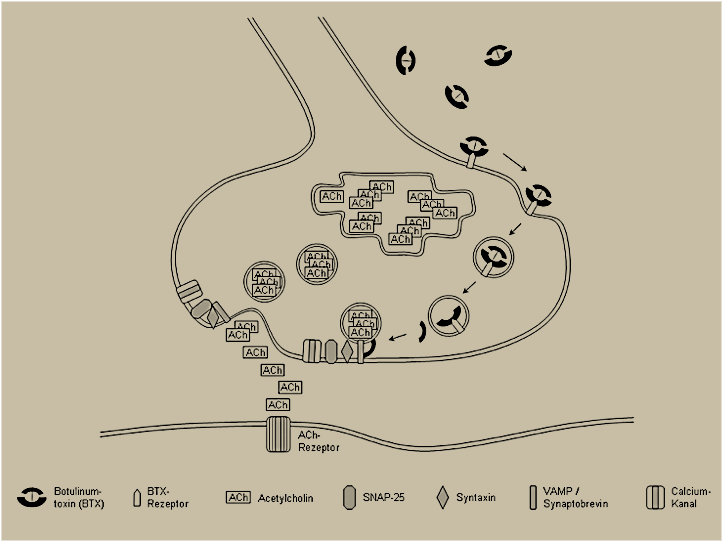
Effect of botulinum toxin A on the nerve endings
Wrinkles
- Due to the targeted weakening of certain facial muscles with botulinum toxin A, wrinkles caused by the movement of facial muscles can be eliminated or at least reduced.
- Botulinum toxin A is particularly suitable for the treatment of forehead wrinkles, frown lines and laugh lines. But it can also effectively treat wrinkles on the nose (“bunny lines”), the upper lip and chin, as well as muscular bands on the neck.
- Whether or not wrinkles can be completely eliminated by botulinum toxin A depends on how long the wrinkles have developed and whether the skin in the valley of a fold has thinned. In this case a full correction can often only be achieved by combining botulinum toxin A with a filler such as hyaluronic acid.
- The smoothing effect on wrinkles is usually achieved after 2–4 days and lasts for 3–6 months.
- Re-treatment is usually required after 6-12 months.
- When started early, botulinum toxin A can completely eliminate facial wrinkles or, with regular use, prevent their occurrence or progression.
Excessive sweating
- Botulinum toxin A is also suitable to treat the condition of excessive sweating of the armpits (axillary hyperhidrosis) and forehead (facial hyperhidrosis), where sweating exceeds what is necessary to normalise body temperature.
- The condition of increased sweating due to disease (infections, cancer, hormonal imbalances, neurological disease) or as a side effect of medication should be ruled out before treatment with botulinum toxin A.
- Excessive sweating often begins in young adulthood, is genetically inherited in 30–50% of cases and can be triggered by emotional stress.
- In severe cases excessive sweating can lead to inflammation and infection of the skin in the affected areas, posing significant psychological stress and potential social isolation.
- Local treatment with botulinum toxin A can alleviate or eliminate these symptoms.
- The duration of the antiperspirant effect of botulinum toxin A varies from patient to patient and depends on the treated skin area and the injection technique.
- The antiperspirant effect occurs after 5–7 days and lasts an average of 6 months.
- Further treatment is usually required after 6–12 months.
- Not every form of excessive sweating can be effectively treated with botulinum toxin. It is possible, though, to surgically remove sweat glands.
Side effects of Botulinum toxin A
- Weakening of muscles or dryness of skin due to reduced sweating.
- A small percentage of patients may experience resistance to botulinum toxin A after many years of treatment. This can be due to the formation of antibodies against botulinum toxin A.
- The immunological reaction of the body to botulinum toxin A and thus the risk of antibody formation depend directly on the protein content of the preparation.
- Clinical studies have shown that even after repeated treatments the use of botulinum toxin A usually remains effective despite the formation antibodies.
Appointments for Botulinum-Toxin A with Dr. Scheufler in Bern
Before any facial treatment you should have a comprehensive consultation and additional examination. Only in this way can you be sure whether the type of surgery will lead to the result you expect. In addition, your consultation will give you a good impression of our experience in the field of facial surgery. Feel free to ask for before and after photos to get an idea of what the results look like.

Dr. med. O. Scheufler, specialist in plastic, reconstructive and aesthetic medicine and author of this website, is pleased to be at your service for a personal consultation. Besides his main work at the Aare Clinic, PD. Dr. Scheufler is also scientifically active and has habilitated at the University Hospital of Basel. He was therefore awarded the title of assistant professor by the University of Basel. In addition, he was honored by Duke University (USA) as a Visiting Professor. So do not hesitate to contact him if you wish a professional consultation!



